LabHQ Q4 Product Update: Introducing Full Stock Management
Highlights from this release
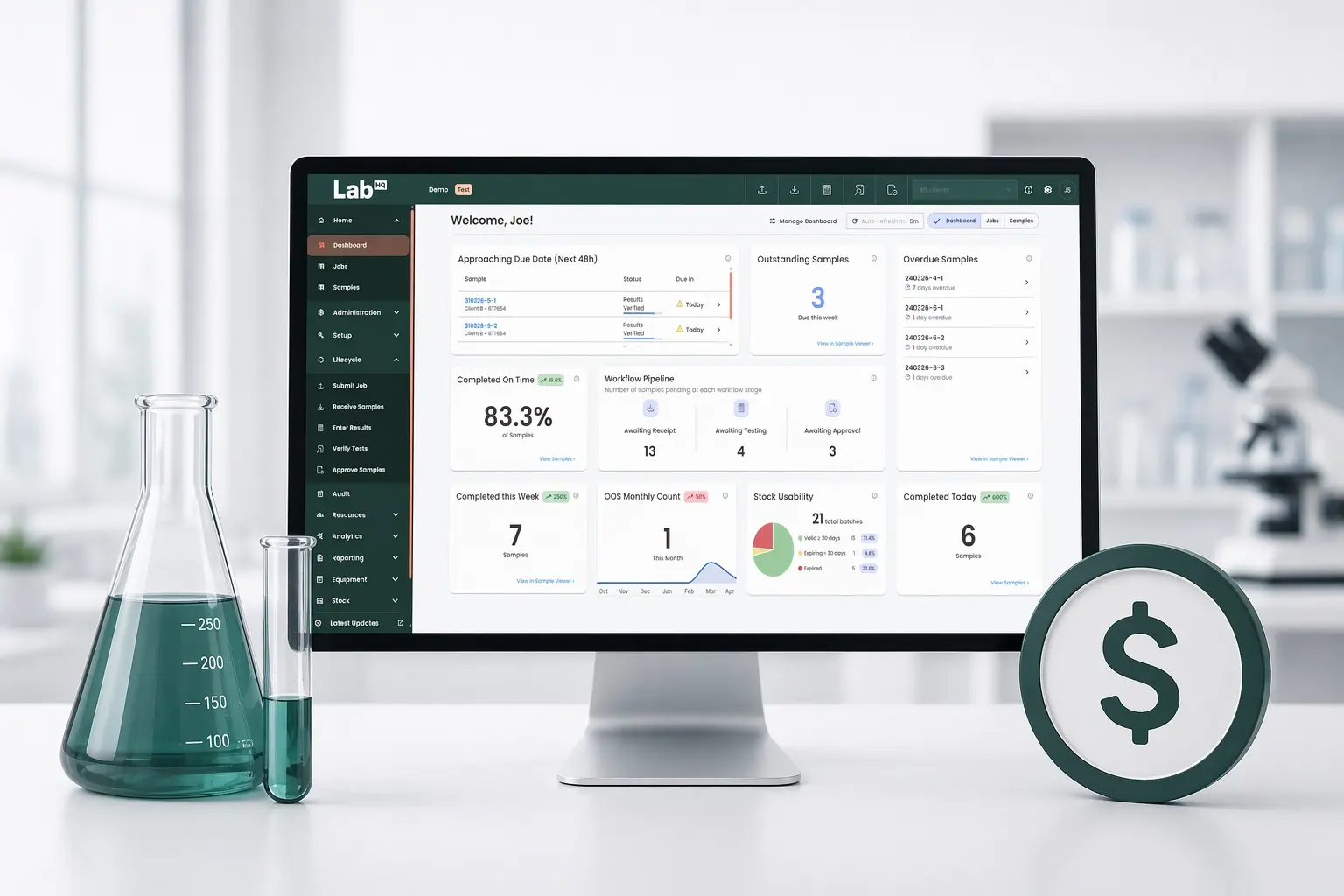
A complete foundation for Stock Management
This release introduces a full set of stock management tools – including templates, batch-level tracking, expiry insights, usage monitoring and integrated QC workflows – giving labs everything they need to track and control stock with accuracy.
What’s new in Q4
- Create and manage stock item templates
Standardise stock setup with predefined templates for item codes, units, minimum thresholds and categories.
Why it matters: Faster booking-in and consistent, clean inventory records.
- Improved stock categorisation
Assign items to categories such as Reagents, Standards, Consumables or Equipment.
Why it matters: Clearer organisation and easier searching.
- Record and track new stock
Record batch number, quantity and expiry when items arrive, with batch records created automatically.
Why it matters: Real-time, accurate stock levels from the moment stock is received.
- Stock inventory summary
See all stock items at a glance, including usable quantities across non-expired batches.
Why it matters: Quick visibility into what’s available.
- Minimum stock thresholds
Set minimum levels per item so LabHQ flags when stock is low.
Why it matters: Helps prevent shortages and supports better planning.
- View and edit batch details
See batch-level quantities, expiry dates and status - and make corrections if needed.
Why it matters: Better traceability and improved data accuracy.
- Batch status tracking
Track batch lifecycle through testing, approval and rejection.
Why it matters: Stronger compliance and clearer quality control.
- Automated QC testing for incoming stock
Trigger QC workflows when batches are received, automatically creating jobs linked to the batch.
Why it matters: Ensures only approved materials enter production.
- Full stock audit trails
All stock actions are automatically logged, including creation, updates, adjustments and usage.
Why it matters: Improved accountability and easier audits.
- Stock adjustments and usage tracking
Record consumption, disposals, corrections and non-testing adjustments - all with reasons.
Why it matters: Accurate inventory levels with complete transparency of changes.
- Expired and expiring stock visibility
See expired batches and those due to expire within 30 days.
Why it matters: Supports better stock rotation and reduces wastage.
- Testing-related stock usage
Link stock directly to test methods and deduct quantities automatically.
Why it matters: Full visibility of what was used in testing and improved traceability.
- Smarter stock search and filtering
Search by item or batch and filter by category, units, users, date range or stock status.
Why it matters: Faster navigation, faster investigations.
- Recently received and recently adjusted stock
Quick access to stock activity from the last seven days.
Why it matters: Helps users stay on top of recent changes.
Curious to see LabHQ in practice?
→ Try LabHQ Essentials free
Additional enhancements
- 2D specification limits
Define two-dimensional tolerance areas with visual pass/fail evaluation.
Why it matters: More accurate assessments for complex specifications like colour tolerance.
Staying up to date
Ready to see what’s new?
If you’re exploring LabHQ for the first time, our quarterly release cycle means you’ll always have access to the latest feature improvements - without disruption or extra costs.
Book a free demo today to see the Q4 updates in action.